Sytse Sijbrandij, the Dutch-born founder of GitLab, has osteosarcoma in his T5 vertebra. After exhausting standard oncology pathways, he is documenting his treatment decisions publicly, publishing 25 terabytes of medical data to Google Cloud, and recruiting other patients into self-directed protocols—converting what would typically remain private medical records into what he frames as a crowdsourced research project.
Dispatch
Amsterdam/San Francisco, January 2025 — Sijbrandij published his cancer journey on his personal website, sytse.com, after running out of conventional treatment options. His statement is blunt about the constraint:
I've taken agency in the treatment of my bone cancer (osteosarcoma in the T5 vertebrae of the upper spine). After I've ran out of standard of care treatment options and there were no trials available for me I've started doing: maximum diagnostics, created new treatments, started doing treatments in parallel, and scaling this for others. [1]
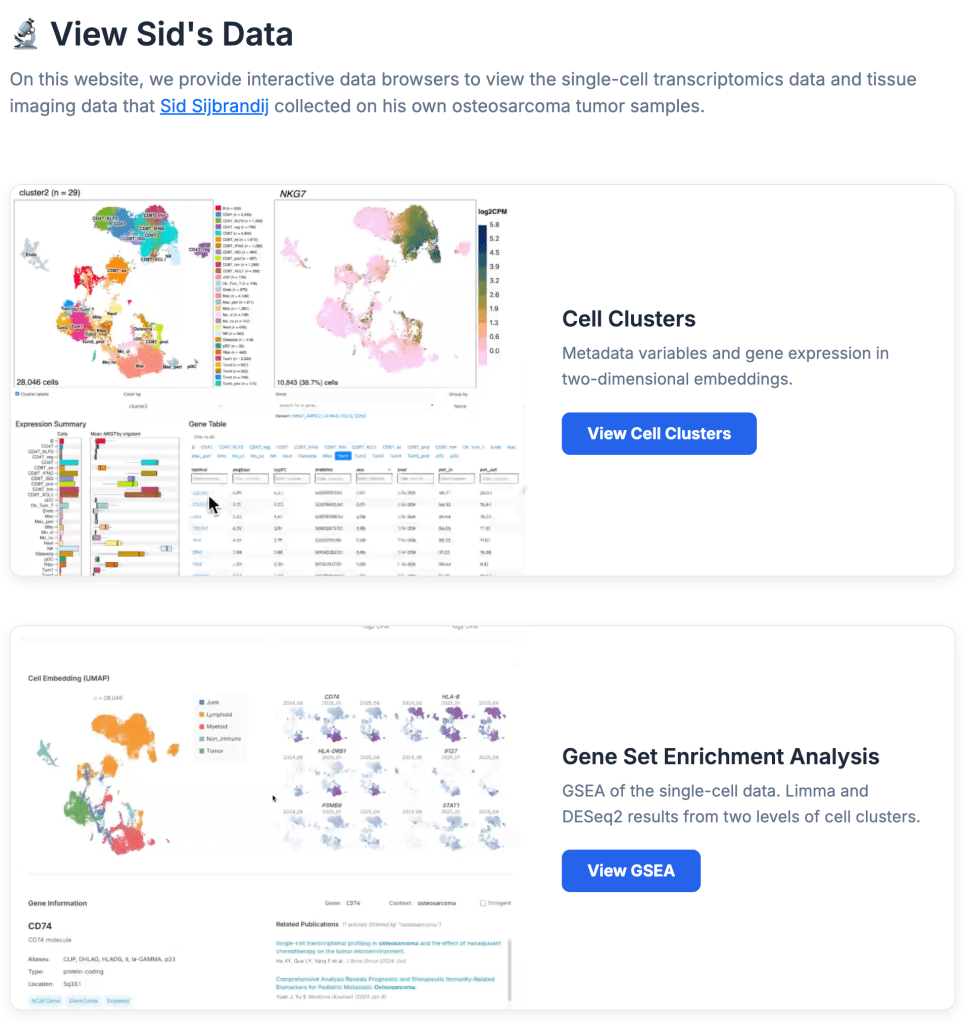
The archive includes a detailed treatment timeline, a cancer journey deck presentation, and a recording of a talk at an OpenAI Forum. He has also created a separate data repository at osteosarc.com, explicitly designed for other patients and researchers to access his medical records. [1]
Sijbrandij frames the move as a critique of medical gatekeeping. He points to an essay by Ruxandra Teslo arguing that regulatory bureaucracy blocks patients from accessing experimental treatments—and he is using his own case as evidence. [1]
No major outlet has yet offered a contrasting account. This analysis draws from the single source above, supplemented by contextual reporting on patient data rights and off-label treatment access.
What's Really Happening
The Real Stakes
For Sijbrandij personally: He is betting that speed and data transparency can outrun a disease that typically progresses quickly. Osteosarcoma in the spine is particularly aggressive; five-year survival rates hover around 70 percent for localized disease but drop sharply once metastasis occurs. [5] By running parallel treatments and publishing his responses in real time, he is attempting to compress the feedback loop—traditional clinical trials take 5–10 years; he is operating on a months-long timeline.
For patient advocacy: Sijbrandij's move will likely accelerate the broader conversation about patient data rights and the regulatory barriers to experimental treatment access. Organizations like the FDA have signaled openness to real-world evidence and patient registries, but they have not endorsed patients running their own treatment protocols without institutional oversight. [6] If Sijbrandij's approach yields a documented remission or extended survival, it will become a case study for patient-led medicine advocates. If it fails, it will be cited as a cautionary tale about the risks of abandoning established oncology protocols.
For GitLab and the tech industry: Sijbrandij has not stepped down as CEO (GitLab remains a private, closely held company). His public cancer fight will likely reshape how tech founders navigate health crises and public disclosure. The move is also a statement about founder agency—he is not hiding his diagnosis or outsourcing his medical decisions to specialists. This carries reputational risk (if the public experiment fails) and potential upside (if it succeeds, he becomes a model for founder resilience).
For medical regulation: The case tests whether the FDA's tolerance for patient-directed research extends to a high-profile founder with resources and media reach. Regulatory agencies typically move slowly on novel approval pathways; Sijbrandij is moving fast. If his data becomes valuable to other researchers or if his treatment decisions produce publishable outcomes, pressure will mount on regulators to formalize pathways for patient-led data collection and parallel treatment protocols. [7]
Industry Context
Osteosarcoma treatment has remained largely unchanged for 40 years. The standard protocol—neoadjuvant chemotherapy, surgery, adjuvant chemotherapy—produces five-year survival rates of approximately 70 percent in developed countries but shows minimal improvement over recent decades. [8] This stagnation reflects the disease's rarity (fewer than 3,000 cases annually in the US and Europe combined) and the limited commercial incentive for pharmaceutical companies to develop new therapies.
Patient registries and real-world data initiatives have become more common since 2015, particularly in oncology. The FDA's 21st Century Cures Act and subsequent guidance documents have explicitly encouraged the use of real-world evidence in regulatory decision-making. [9] However, these registries typically operate under institutional oversight—hospitals, universities, or non-profits manage the data collection and analysis. Sijbrandij's model—a single patient publishing raw data and inviting others to participate—is more decentralized and less precedented.
The GitLab founder is also tapping into a broader tech-sector interest in healthcare disruption. Companies like Tempus, Flatiron Health, and Color Genomics have built billion-dollar businesses by aggregating and analyzing cancer patient data. [10] Sijbrandij's approach is lower-tech but more transparent—he is publishing data directly rather than licensing it to intermediaries.

Impact Radar
Watch For
1. FDA or EMA response: If either regulator issues formal guidance on patient-directed treatment protocols or real-world data collection by Q3 2025, it signals that Sijbrandij's case has moved from novelty to policy consideration. [No public timeline established.]
2. Clinical trial enrollment: Monitor whether other osteosarcoma patients attempt to replicate Sijbrandij's parallel-treatment model or publish their data publicly. A cluster of similar cases would indicate a shift in patient behavior and could pressure oncology institutions to formalize patient-led research pathways.
3. Sijbrandij's health trajectory: His public updates on treatment response will become the primary evidence base for whether the approach works. If he publishes data showing tumor regression or extended survival beyond historical benchmarks, the model will gain credibility; if progression accelerates, it will be cited as evidence that parallel treatments outside institutional oversight carry additional risk.
4. Regulatory enforcement: Watch for whether the FDA or state medical boards take any action against physicians treating Sijbrandij or other patients using the osteosarc.com protocol. Regulatory forbearance would signal tacit acceptance; enforcement would indicate institutional resistance.
Bottom Line
Sytse Sijbrandij is not the first terminally ill patient to seek alternative treatments, but he is the first with a $6 billion company, technical infrastructure, and a public platform to scale that impulse into something resembling a research collective. Whether his approach succeeds or fails, it will force regulators, medical institutions, and tech companies to confront a question they have mostly avoided: What are the conditions under which patients should be allowed to run their own medical research? The answer will reshape how rare-disease communities access experimental treatments and how medical data flows between patients and institutions.
